Industries#nonfoods0DTE2023 trends2024 holiday season202624/7 trading3AF3M4As7 Mindsets80/2080/20 ruleAAPIAASAAbercrombABMAboutabsenteeismAcademic integrityACCaccelerated learningacceleration-based curriculumaccess to careaccessibilityaccessibility analysisaccount-based marketingaccountabilityachievementAcostaacquisitionsad targetingAdam BehrensAdam BernardiAdam RosenADASAdena FriedmanadhdadministratorsAdobeadolescentadsadtechadverse eventAdvertisingadvertisingadvertising agencyAdvisory Boardadvisory boardsAFCOMaffordability crisisaffordableaffordable broadbandaffordable care actaffordable connectivity planageismageistAgencyagentic AIagileagreementagricultureahipAHIP 2023AHIP 2024ahip 2025AIAI adoptionAI and machine learningAI chatbotAI data centersAI governanceAI ImpactAI Impact SummitAI Innovation AwardsAI LawAI literacyAI sentimentAI stewai toolsAI translationAI trendsairlinesairportairport solarAlbertsonsalcohol use disorderalcohol-free beveragesAlex HonnoldAlex PaglianoAlfred JohnsonalignmentallergensAllister HercusAllUnite.comalternative transportation solutionAlun GreenAmazonAmazon AdsAmazon Prime DayAmbreen AliAMC NetworksAMDAmerican AirlinesAmerican Cancer SocietyAmerican Chemistry CouncilAmerican Clean Power AssociationAmerican Culinary FederationAmerican Public Health AssociationAMLAmperonAmplify Desmos MathanchorAngela HaynesangeranimeAnna BagerAnnual Meat Conferenceannual training conferenceAnthony AttiaAnthropicanti-fraudAntronixanxietyApolo.usappleapprenticeshipappsAPQCararcharchitectArgonne National Laboratoryart of leadershipArt Pettyartificial intelligenceartificial intelligence coachingAsaf GreinerASCDASCEAscensionASDAsiaAspiration Economyassessmentassessmentsasset classesassistive technologyat-risk studentsAT&TathleticathleticsAtlantic Shores WindATSCAudiaudience engagementAuditBoardaugmented realityauthenticityauthorauthorityautismautomated complianceautomationautonomous drivingautonomyAVAVAvi Chai OutmezguineAviation & AerospaceAviation & AerospaceAzoyaB CorpB2BB2B content marketingb2b marketersb2b marketingB3back to schoolBank of Englandbarbara corcoranbariatric surgeryBattery Council Internationalbattery energy storagebattery metalsbattery storageBCIBCN Visualsbehaviorbehavioral changebelongingben & jerry'sBESSbest of breedbetter leadershipbetter learningbetter-for-youbeveragebeveragesbiasBidenBig Beautiful Actbig beautiful billBig Beuatiful BillBig ShowBig TechBigCommerceBill GulyaBill Kleymanbillboardbillboardsbiotechbirthbitcoinbitcoin futuresBlack communityBlake Lawrencebleisurebleisure travelblinking 12blockchainBloombergNEFBoathouse agencyBoca50body dysmorphiabody languageBonnie Chanbrainstormbrandbrand awarenessbrand identitybrand lovebrand loyaltybrand marketersbrand marketingbrand purposebrand safetybrand strategybrandsBrazilian realbreast cancerBrene BrownBrian Niccolbridgebridge to bankabilityBrightcore EnergybroadbandBroadpeakbrowser cookiesBSCbudgetbuildersbuildingbuilding better career successbuilding communitybuilding connectionsbuilding effective teamsbuilding relationshipsbuilding resiliencebullyingbundlingburnoutBurns & McDonnellbusinessBusinessBusinessbusiness email compromisebusiness financebusiness modelbusiness operationsbusiness planbusiness regulationsbusiness strategybusiness transformationbusiness travelbuyingBYOPc-suitecablecable operatorcable standardscalculatorCalifornia Resources CorporationCanadian Prosthetics & Orthotics Journalcancercandycannabiscarcarboncarbon emissionscarbon emissions offsetscarbon offsetscareercareer advicecareer and technical educationcareer developmentcareer growthcareer planningcareerscarpentryCboeCBPcbscbtCCPsCDCCDNcell-based meatcentral bank futuresCEOcertificateCES 2026CFOCFTCchallengechangeChange Healthcarechange managementCharlotte AliottCharlotte AlliotchatbotsChatGPTchefschemicalCherry Street Energychild carechildren with disabilitiesChinaChklovskichoice mapchoice overloadChris CreedChris EdmondsChris RhodesChristensen InstituteChristine Larsonchristmaschronic absenteeismCHROschunkingCignacineverseCiscoCity of Atlantacivicscivil engineerclarityclassdojoclassroomclassroom controlclassroom designclassroom heatclassroom lessonclassroom managementclassroom technologyClaude Opus 4.5ClawdbotcleaningCleanpowerclearing processclient segmentsclimate capitalclimate changeclimate disclosureclimate riskClimate Weekclinicianscloudcloud computingclubsCMARCME GroupcmoCO2coachingcoffeecoffee-badgingcognitiveColgate-Palmolivecollaborationcollegecollege counselorColliersColumbuscommand-and-controlcommencement speechcommercecommoditiescommodities marketsCommodity Futures Trading Commissioncommunicationcommunicationscommunitycommunity collegecommunity resourcescommunity solarcommutecompany culturecompany purposecompany valuescomparisoncompassioncompetencycompetitioncompliancecompositioncompoundingCompTIAcomputercomputer sciencecomputer trainingconcreteconferenceconferencesconflict managementconflict resolutionconnected TVconnectionsconnectivityconscious brandsconsolidated platformsconstructionconsumer choiceconsumer insightsconsumer packaged goodsconsumer privacyConsumer Productsconsumer trendsconsumer valuescontent marketingcontinuing educationcontinuous controls monitoringcontinuous improvementcontractconvenience storesconventionconvergenceconversationCool EffectCOP26COP27COP28copilotcord cuttingcore valuescorporate boardscorporate culturecorporate debtcorporate financecorporate social responsibilitycorporate strategycorporate volunteerismcost of capitalcostcoCouncil of Chief State School OfficerscourageCoveocovidCOVID-19COVID-19 pandemiccoworker connectionsCPGCraig McDonoghcreativitycredit flowcredit spreadscrisis communicationscrisis managementcritical carecritical thinkingcriticismCRMcross-border connectivitycruisesCruxCrux Climatecryptocrypto ETFscrypto ETPsctamCTECTVcultural considerationscultural trendsculturally inclusivecultureculture codecuriositycurriculumCurriculum AssociatesCustomer Engagementcustomer experiencecustomer experience strategycustomer loyaltycustomer problemcustomer relationship managementcustomer servicecustomer trustcustomer-centriccustomerscyber attackcyberattackcyberriskcybersecuritycybersecurity regulationscyberthreatcyberthreatsDaniel Cruisedaniel golemandatadata analyticsdata centersdata governancedata managementdata marketingdata privacydata sciencedata servicesdata silosDataBeeDatassentialDavid Frumdaylightingdebatedecarbonizationdecencydecision-makingdecisionsDeeper LearningdeepfakesdefensedegreeDEIdelegationdeliveryDeloitteDelta Air Linesdemocracydental insurancedental networkDepartment of EnergyDerek Sammannderivativesdesigndesign thinkingdestinationsdetroit tigersdevelopmentdevicesDéVon Christopher Johnsondiabetesdiagnosisdiagnosticsdialoguedifferentiationdifficult peopledigitaldigital advertisingdigital dividedigital front doordigital literacydigital marketingdigital mediadigital pathologydigital storefrontdigital toolsdigital transformationdigital TVdigital twindigital validationdigitalizationDirect paydisabilitiesdisabilitydisciplinary actiondisciplinedisease detectiondisgruntled workersdisruptiondistributed teamsdistributiondiversityDMISTdocsisDOCSIS 4.0document management systemdomestic contentdomestic manufacturingdonationDonna SpanglerDOOHDouyinDr. Martin Luther King Jr.drone attacksdrug developmentDrug Enforcement Administrationdrug policydrug pricesdunbarDVBdyslexiae-booke-commerceE-liabilityE-ratesearly childhoodearly childhood educationearly detectionecommerceeconomic developmenteconomicseconomyEDF power solutionsEDF Renewablesedtechedtech toolsEducationEducationEducationEducationeducationeducation cybersecurityeducation leadershipeducation technologyeducational coachingeducational leadershipeducatorsEdward Decieffective communicationefficienciesefficiencyEHRELAelectric vehicleelectricianselectronic health recordselectronic trading solutionselementary schoolElisabeth HayesEllen Beckelon muskemailemail marketemail marketingemail segmentationemerging leadersemerging market currenciesemotional integrityemotional intelligenceemotional wellbeingemotionsempathyEmplifiemployeeemployee behavioremployee coachingemployee communicationemployee conflictemployee developmentemployee empowermentemployee engagementEmployee Experienceemployee fulfillmentemployee growthemployee loyaltyemployee motivationemployee performanceemployee productivityemployee recognitionemployee recruitmentemployee retentionemployee stressemployee trainingemployee trustEmployee Well-beingemployeesemployees and culture changeemployees of coloremployer benefitsemploymentempowermentENCOREencryptionenergyEnergy & ChemicalsEnergy & Industrialenergy community creditsenergy demandenergy efficiencyenergy forecastingenergy management systemsenergy marketsenergy portfolioenergy securityenergy storageenergy transitionengagementengineerengineeringEnglishenglish language learnersEnglish-language learnerEnterpriseentertainmententrepreneursenvironmentenvironmental liabilityequityERCOTergonomicsEsben ElmoeESGESLespnesportsEssenceMediaComEthereumethical marketingethicsEuronextEV chargingevent marketingeventsEventsEverDrivenevolving managerexcellenceexecutive coachingexecutive functionexecutive presenceexpertexposed dataextended spectrumextra creditEYfacebookfacility managementfamiliesfamily engagementfanbaseFancy Food 24/7FASBfatFCAFCCFDAfear of failureFederal Reservefeedbackfemale leadersFEOCFERCFIAFIA Bocafiberficofield tripFilling in the Blanksfilmfinal ruleFinancefinanceFinanceFinancefinancial market infrastructurefinancial technologyfintechfirst-party dataFlaviaflavor trendsflexibilityflexible workflexitarianfloating windflowFluenceFMFMIfocusfogfoodFoodFood & BeverageFood & Beveragefood allergiesfood and beverage trendsfood inflationfood insecurityfood retailfood retailersfood retailingfood safetyfood technologyfood trendsfood wastefoodservicefootballforecastForeign Entities of Concernforeign languageformerly incarceratedforresterForrester Consultingfortnitefranchise businessFrank BelzerFranklin ChuFred EndeFred Ledleyfree programfresh foodfresh foodsfrozen foodfrozen foodsFTSE RussellFTXfull duplexfunctionalfunctional beveragefurniturefuturefuture of workfuturesfutures and optionsFutures for KidsFutures Indsutry Associationfutures industryFutures Industry AssociationFX businessgamesgamificationgamified learninggaminggas turbinesGaskellgastroenterologistGDPRgeminigen aiGen ZGenAIgendergender pay gapGene KernsGeneration Zgenerational dividegenerative AIgenerative questionsgeofencingGeography of Brand Connectiongeothermalgeothermal energyGFANZGHDGHGGiada De LaurentiisGies College of Businessgifted and talentedgirlsgirls and STEMGithubGlobal Business Travel Associationglobal flavorsglobal marketsglobal powerglobal tech trendsGLP-1glucosegoal settinggoalsgold contractsgolden state warriorsgolfGood Food InstituteGoogleGoogle Cloudgovernment shutdowngracegrade-level contentgradesgraduate certificategraduate degreegraduatesgraduationgrantsgratitudegray zoneGreat ResignationGreater Houston Partnershipgreekgreen hydrogenGreen NinjagreenwashingGreg AndersonGreg BoyleGreg Garciagrid resiliencyGrid StrategiesgriefgrievanceGriffith Universitygrocersgrocerygrocery appsgrocery retailgrocery shoppinggrocery technologyGroupMgrowthgrowth mindsetguidance counselorguided processGuillaume DeschampshabitshackershackinghallmarkHanger ClinichappinessHarris Pollhatredhbcuhealthhealth and wellnessHealth Carehealth careHealth Carehealth care accesshealth care affordabilityhealth care costshealth care cybersecurityhealth care executivehealth care fraudhealth care marketinghealth care policyhealth care providershealth care techhealth care workforcehealth crisishealth equityhealth insurancehealth planhealth planshealth riskHealth Sector Coordinating Councilhealth systemhealth system Endeavor HealthHealthcarehealthcarehealthcare innovationhealthcare technologyHealthEquityhealthIThealthyhealthy foodhearingHeathrow power outageheavy industrialsHenry FordHenry KissingerHHShigh-performing leadershigher edhigher educationhigheredHilton Hotels brandshiringhiring former felonHispanic marketinghistoryholidaysholisticholistic carehome brandsHomeboy Industrieshomelesshomeless studentsHong KonghopeHoracio Gavilanhorizontal thinkingHorowitz Researchhospitalhospitalityhospitalshot spotshotelhotelshousingHouston Energy Transition InitiativeHoward BeharhrHuloophuman bodyhuman leadershiphuman resourceshumorhungerHuntresshurricaneshybridhybrid eventshybrid workhybrid workerhydrogenHyerhype cyclehyper-targetinghyperscalersiabICEICE ClimateICE Futures EuropeICE MarketsICHICMAICUICYMIidentityidentity verificationIDXiiotIkeaillegal drugsillnessillustrative mathiMBAimmigrant studentsimmunityImpact Cycleimpatienceimposter syndromimposter syndromeimpostor syndromeimprovementimprovisingin-game marketingincivilityinclusionincrease teamworkindoor farmingindustrial iotIndustrial Solarinfancyinfant healthinfectioninfection controlinfection preventioninflationInflation Reduction Actinfluenceinfluencer marketinginformation technologyInfrastructureInfrastructureInfrastructureinfrastructureingredientsinnovationinspirationinspiring othersinstagramInstitute of Medicineinstitutional investorsinstructioninstructional coachinstructional coachinginstructional practicesinsuranceinsurance crisisInsurance Institute for Highway Safetyinsurance premiumsinsurerinsurersintegrityintelligent transportationInteractive TVinterconnectioninterconnection queuesIntercontinental ExchangeIntercontinentalExchangeIntercontinential Exchangeinterest ratesInternational Derivatices ExpoInternational Derivatives ExpoInternational Equal Pay DayInternational Literacy AssociationinternetinteroperabilityinteroperabiltyinterventioninterventionsIntrenational Derivatives Expointrovertsinvention educationinvestinvestment tax creditinvestoriotIouri SaroukhanovIPTVIsabel KunkleISPEITIT supportIT workflowITCJackie WolinJacob LaneJames DiceJamie GambleJane StrickerJason KeehnJason PeartJawsJB MackenzieJean-Pierre LacroixJeff SprecherJeffrey JukubiakJeffrey PfefferJennifer GranholmJennifer HarringtonJenny NicholsonJess MessengerJessica LibbyJessie JohnsonJigar ShahJill BlicksteinJim Lehrerjo hamiltonjob huggingjob marketjobsJohn BaldoniJohn DuganJohn StankeyJohnson ControlsJonah SchenkerJones ActjournalingjoyJoydaysJulie WinklerJustin Thomas-CopelandK-6Karla Fernandez ParkerKarthik RamannaKaryn LewisKate Scott-DawkinsKatie LinfordKeeper SecurityKent LewisKerry LandolfekeynoteKIBOKids food and beveragekilt challengeKim MarkuskindergartenkneeKPMGKPMG USKristin NeffKrogerKYClaborlabor marketlaboratorylabslanguagelanguage learningLaRae QuyLarry LawrenceLarry RobertsonlatinlaughterLauren Collinslawlaw firmslayoffslead generationleadersLeadershipleadershipleadership adviceleadership agilityleadership communicationleadership confidenceleadership courageleadership credibilityleadership developmentleadership effectivenessleadership lessonsleadership resilienceleadership strategyleadership styleleadership successleadership trustleadership valueslearninglearning gaplearning recoveryLegallegal intelligencelegal techlegaltechlegoleisurelessonsLevel 3LGLGBTQLGBTQIAlibrarieslibrarylicensingLife Sciences & TechnologyLife Sciences & Technologylife skillslifelinelifelong learninglifestyle changeslifetimeLightcastlikeabilitylikeableLina MaggiLinkedInliquidity marketsLisa GraystonlisteningliteracyLiumLium ResearchLiveRamplivestream shoppinglivestreamingLoan Programs Officelocation-basedlodginglogisticslonelinesslong-term carelorne michaelslosslow-glycemic ingredientsloyaltyloyalty programloyalty programsLPOLuis SilvaLuiza DemôroM&AMac minimachine learningMaggie Pelosomakerspacemalwaremanagementmanagement stylemanagersmanaging a teammanaging upmanufactured foodmanufacturingMarc Cugnonmargin efficienciesmarijuanaMark McClellanMark Reinboldmarket integritymarket researchmarket volatilitymarket volumesmarketersMarketingmarketingMarketing & AdvertisingMarketing & Advertisingmarketing agenciesmarketing automationmarketing best practicesmarketing budgetmarketing careermarketing jobsmarketing strategymarketing trendsmarriottMarshall GoldsmithMary Ann O'Brienmaster'smaster's degreematernal healthmaternal morbiditymathmath anxietymath educationmath manipulativesmathedMatt FerrellMatt JudgeMBAMcDonald'sMcKinney-VentoMD Anderson Cancer Centermeal planningmeasurementMeat Institutemeat-buying trendsmediaMedia & Entertainmentmedia planmedia relationsMedicaidmedical marijuanaMedical Product Manufacturer Cyber Incident Response PlaybookMedicareMedicare AdvantagemeditationMedTechmeetingsmenopausemental healthmental health caremental toughnessmentormentoringmentorsmentorshipMenu - Mainmenu innovationmenu trendsmenus of changemergersmeritocracymessagingmetaMeta data centersMetaversemetricsMichael ChaninMichael EppolitoMichael GaskellMichelle CollinsmicrocredentialsmicrogridmicrohabitsmicromanagementmicroplasticsMicrosoftmiddle managersmiddle schoolmidmarket wholesale distributorsMike Dahermike driehorstMike RichtermillennialsmindfulnessmisalignmentmisinformationmismanagementMISOmissileMistubishi Powermixed-useMobbie Nazirmobile gamesmobile marketingmocular technologyModern Energymodular designMoltbotmonetizationmoral injurymoral leadershipmortgage-backed securitiesmotivationmotivation sciencemoviesMTSSmulti-asset exchangemulticultural marketingmultigenerational workforcemuseumsmusicMyers-BriggsMyles Washington IIImythNAB ShowNAEPnarcissismNASSPNational Academy of MedicineNational Education SummitNational Renewable Energy LaboratoryNational Restaurant AssociationNational Restaurant Association ShowNational Teacher of the Yearnatural & organicnatural and organicnatural disastersnatural language processingnature playNAWnbcnegotiationneruosciencenet energy meteringnet zero as a servicenet-metered solarnet-zeroNetDocumentsnetworknetwork accessibilitynetworkingneurodiversitynew yearnewcomersnewfrontsnewslettersNextGenNexus LabsNexusConNFTsNHCAANicole PennNIHnonfoodnonfungible tokensNRELNRFNRF2026NTIANumeratornursenurse practitionernurse scientistnursesnursingnutritionNvidiaNWEAOAAAobesityObesity Action Coalitionofficeoffice perksoffshore windOld Westolder workersOliver MetcalfeomnichannelonboardingOne Big Beautiful Billone-on-onesonline continuing educationOnline gamingonline groceryonline MBAonline schoolonline sellersonline shoppingOOHOOH advertisingOpenAIoperationoperational technologyoperationsopioid epicemicoptimism biasoptionsorganizational cultureorthoticsOtter PRout of homeout of home advertisingOut of Home Advertising Association of Americaout of networkovercoming adversityoverrunpackaging innovationpackaging/labelingpancreatic cancerpandemicparaprofessionalsparent communicationpartnering on volunteeringpassionpassword managementpasswordspatentpatient capitalpatient carepatient experiencepatient portalpatient safetypatientsPaul Woodspayerspayment integritypaymentspediatricianpediatricianspeer connectionspeer supportpeople developmentpeople-pleasingpepsicoperceptionperinatal quality collaborativespermissionperpetualspersonal assistantpersonal developmentpersonal growthpersonality typespersonalizationpersonnel vs. personal decisionspet foodPeter DruckerPeter Prodromoupharma manufacturingpharmaceuticalpharmaceuticalsphilanthropyphilosophyphishingphonesphonicsphysical AIphysicianphysicianspizza hutPJMPlant Based World Conference & ExpoPlant Based World Expoplant-basedplant-based foodplant-based foodsplant-based innovationPlant-based meatplant-based proteinplaybookPLC modelPLCsplumbingpodcast advertisingpodcastspoetrypolicypoliticspop-up adspop-upsPorschepositive energypositivitypostpartumpower gridPower KneePower of Meatpower purchase agreementsPowerFlexPPAspraisepredictionprediction marketspregnancypreparationpreschoolprescription drug costsPrescription drugsPresentation C.O.D.EPresentation tipspresentationspressurepretermprice discoveryprimary careprincipalPrincipal of the Yearprior authorizationprivacyprivate-labelprivileged access managementProactive Availability ServicesProactive Problem Managementproblem-based learningproblem-solvingprocessed foodprocurementproduct developmentproduction tax creditproductivityprofessional developmentprofessional etiquetteprofessional learningprofessional learning communityprogrammatic adsprogrammatic advertisingproject managementproject-based learningpromotionprosthesesprosthesisprostheticsProtein PACTprovider accessibilityproviderspsychological safetyPTCpublicpublic acceptancepublic healthpublic policypublic relationspublic speakingpublic transitPublicationspublishersPublixpuerto ricopunctualitypurposepurpose marketingPwCq&aQLCQR codeQR codesquakequalificationsquality feedbackquality improvementquan zhangquantum computingQuantum Corridorquantum innovationquestionsquiet quittingRabbit R1railRaj NijjerRandy KrosznerransomwareRashan DixonRazerRE+Readers' Choice Awardsreadingreal currency futuresreal estateReal Estate Wealth Networkreal-time tradingrecessionreconciliation billrecruitmentRedditregenerative agricultureregulationregulationsregulatory approvalreinventing educationrelational influencerelationshipsremoteremote monitoringremote workremote workerrenewable energyrenewablesrepo clearingrepo expansion initiativeRepresentative Dan Meuserresearchresidential solarresilienceresiliencyresilient staffresist changeresolutionsrespectrestrestaurantrestaurant menurestaurant technologyrestaurantsretailRetailRetailRetailretail growthretail marketingretail mediaretail participationretail technologyretail tradingRetentionretirementretiringretreatreturn to the officerevenuerewardsRichard RumeltRick Milenthalrigidityriskrisk appetiterisk managementRivianroadsRob GramlichRobert Downey Jr.Robinhoodrobloxrobotaxiroboticsrobotsrock climbingRodrigo BenaventuraRoger MiksadroundupRSACruralrural carerural healthrural health carerural hospitalsRussell 2000Russia/Ukraine warRyan ClareRyan FitzgeraldRyan LaflerRyan PfaffS. Chris EdmondsS&P 500SaaSsabbaticalSAESAFSafety TechnologySage GeosystemssalarySalesforceSally HelgesenSamsungSAPsaturated fatsaturday night livesaying nosbeducationSBTiscammersscheduleSchedule ISchedule III drugschoolschool attendanceschool climateschool cultureschool districtschool environmentschool leaderschool leadersschool librariesschool missionschool safetyschool safety measuresschool transportationschool violenceschool visionschoolssciencescience of readingScope 3scott harrisScott MorrisScout Clean Energyscreen timeSDI PresenceSDOHSean Kellysearch engine marketingseaweedSECSecOpsSecurities Idustry and Financial Markets Associationsecuritysecurity data risk fabricsecurity policySELSEL successself-awareself-awarenessself-checkoutself-compassionself-confidenceself-drivingself-hosted AIself-promotionself-servicesellingsenior leaderssensorySEOservant leadershipServiceNowset-jettingsex edSGXshameshareholder capitalismshark tankShawn CreightonSheila MorinShell New EnergiesShelly PalmerShipAccelShippingshipping solutionsshootingshopper journeyshort-form videosshowsSIFMAsilencesimon & garfunkelSingapore ExchangeSingles Dayskepticsleepslow lookingsmall businesssmart buildingsmart buildingssmart cartssmart citySmart City Challengesmart homesmart ringssmartbriefSmartBrief Innovation AwardsSmartpulseSmartSummitSmithsonianSmithsonian Institutesmoke-freesnackssnapSNLSnorre Kjesbusocialsocial commercesocial commerce and retail mediasocial determinants of healthsocial engineeringsocial mediasocial media habitssocial media marketingsocial studiessocial-emotionalsocial-emotional learningsodiumsoft skillssoftwaresolarsolar eclipsesolar energysolar glaresolar powersolarSATsolaSATsolid state drivesSolidigmSonySoutheastern GrocersspaceSpanglerSpanishspecial edspecial educationspecial needsSpecialty Food Associationsponsorsponsored accesssportsspotifySSDsstable coinsstaffstakeholdersStar ratingsstarbucksstate of the data centerSTEAMSTEMsteph curryStephanie RodriguezSteve RosaStill to Be Determined podcaststoicismstoicsstore managerStore Manager Awardsstorytellingstrategic planningstrategyStrawberryFrogstreamingstreaming adsstressstress managementStuart Williamsstudentstudent achievementstudent behaviorstudent choicestudent experiencestudent healthstudent mental healthstudent performancestudent stressstudent successstudent supportstudent transportationstudentssubscriptionssubsidiessubstance use disordersubstitute teacherssuccess skillssuccession planningsugarsummer campSummer Fancy Food ShowSummer MersingersuperSuper BowlSuper Bowl adssuperintendentsuperintendent of the yearsupply chainsupportsupportsSustainabilitysustainable aviation fuelSustainable Financesustainable infrastructuresustainable packagingSuzanne LetaSweets & SnacksSwoonswotSyed BalkhiSystem1T+1talenttalent acquisitiontalent pipelineTargettargetingtarifftariff majeuretariffstasktax billtax creditstax incentivesTaylor Swiftteacherteacher burnoutteacher coachingteacher of the yearteacher shortageteacher stressteacher supportteacher workforceteachersteachingteamteam leaderteam softwareteamworkTechTechTech & Learningtech edtech policyTech TipTech Tipstech tolerancetechnical debtTechnical EducationTechnologytechnologytechnology tools.technology-AITechnovationTechTipTed LassoTEDxteenstele-icuTelecomTelecomtelehealthtelemetrytelephoneTeleste InterceptTErry DuffytestingTexas A&MTHCThe Associated General Contractors of AmericaThe Big LebowskiThe Culinary Institute of AmericaThe Food Industry AssociationThe Gallup OrganizationThe Great ResignationThe Hartman GroupThe National Confectioners AssociationThe National Restaurant AssociationThe Pew Charitable TrustsThe Shipyardthought leadershipthreat actorstiktoktimetime managementtimelineTitle IXTiVoTogal.AItokenizationtokenized collateralTom Preston and Luiana NuñeztoolsToshiba Global Commerce Solutionstourismtoxic customerstoxic leadertoxic positivityTPACKtraceabilityTracie Swilleytrade and customstradestradingTrading Technologiestrading technologytraditiontraffictraffic safetytrainingtraitstransferabilitytransferable tax creditstransittransitionstransmissiontransparencytransporttransportationtransportation safetyTraveltravelTravel & HospitalityTravel & Hospitalitytravel marketingTravis KelceTreasury clearingtrendsTrumpTrump administrationTrump majeuretrusttrust managementtutoringtvtv specialstwitterTyler BishopUkraineuncertaintyUndecided with Matt FerrellUnileverUnited AirlinesUnited HealthcareUnitedHealthuniversal service funduniversitiesuniversityUniversity of South CarolinaunpluggingupskillingurbanUS Bureau of Labor StatisticsUSA Batteries Actuser experienceUSFutility-scale solarV&Evacationvaccine hesitancyValentine's Dayvaluevalue-based carevaluesVance MerollaVantaveganvegetarianvehiclevertical farmingveteransvideo coachingvideo gamesvideo marketingvideo productionViktor FranklVinson & Elkinsviolencevirtual diagnosisvirtual learningvirtual realityvirtual teachingvisionVision Provisual impairmentvisual novelsvocabularyvoluntary quitsVRvulnerabilityWade GungollWall-to-Wall maintenance servicesWalmartWalt LukkenWalt Musialwaterwearablesweather derivatives.weatherizationweaving influenceweb designweb developmentWeb3weight lossweight loss drugswell-beingwellbeingwellnessWhen Growth Stallswhiteboardwhole childwholesalerWi-Fiwildfireswind energywind turbineswinewomenwomen in leadershipwomen's healthwomen's sportsWonderhellWood MackenzieWoodMAcwork culturework-life balanceworkforceWorkforceworkforce developmentworkforce trainingworkloadworkplaceworkplace communicationworkplace cultureworkplace technologyWorlds of Flavorwritingwrong-way drivingWWDxrYama Darrietyouth mental healthyoutubeZeliszero trust
Sponsored content
Higher Education Leadership
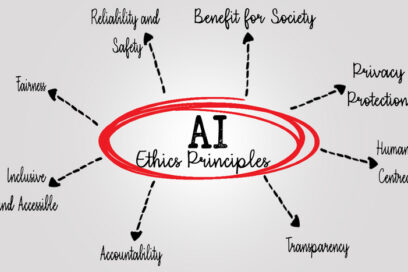
Ethical, human-centered AI is a leadership skill

AI Leadership
“How To AI” author Christopher Mims makes AI approachable for business leaders

Consumer Insights CPG
How retailers can make the most of the feline phenomenon

Consumer Insights CPG
Bridging the nutrition gap with shelf-stable staples

Development Education
Managing AI, not just using it

Food Restaurant and Foodservice
Solving the QSR consistency challenge in high-pressure kitchens

Finance Leadership
Small business cash flow: Visibility, volatility and the road to financial confidence

Consumer Practical AI
AI in everything, everywhere, all the time

Practical AI Technology
Beyond burnout: Building modern SecOps with Agentic AI

Technology
Achieving the full scale of Agentic AI

Security Technology
Getting the right security in place for Agentic AI

Edtech Education
The role of edtech in supporting powerful learning